Latest Research News
-
73
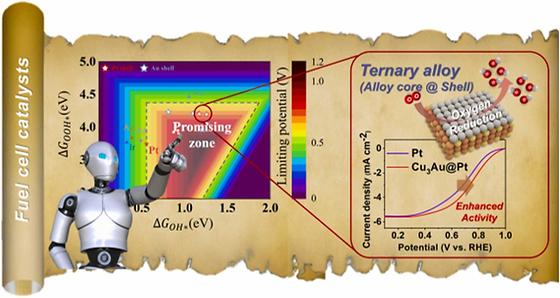
Using AI to develop hydrogen fuel cell catalysts more efficiently and economically
- Development of a new ternary alloy (Cu-Au-Pt) catalyst that is cheaper and more efficient than traditional platinum (Pt) catalysts Proton exchange membrane hydrogen fuel cells (PEMFCs) used in hydrogen vehicles use expensive platinum catalysts to facilitate the oxygen reduction reaction at the anode. There are a vast number of elemental combinations and compositions that need to be explored to develop more efficient and cost-effective catalyst materials than platinum, and researchers are still doing a lot of trial and error in the lab. The Korea Institute of Science and Technology (KIST, President Seok Jin Yoon) announced that Dr. Donghun Kim and Dr. Sang Soo Han of the Computational Science Research Center, Dr. Jong Min Kim of the Materials Architecturing Research Center, and Prof. Hyuck Mo Lee of the Department of Materials Science and Engineering at the Korea Advanced Institute of Science and Technology (KAIST, President Kwang Hyung Lee) have presented a new artificial intelligence-based catalyst screening methodology and succeeded in developing a new catalytic material based on a ternary element-based alloy (Cu-Au-Pt) that is cheaper and performs more than twice as well as pure platinum catalysts. [Figure 1] GRAPHICAL ABSTRACT OF MACHINE LEARNING-DRIVEN HYDROGEN FUEL CELL CATALYST DESIGN The team developed Slab Graph Convolutional Neural Network (SGCNN) artificial intelligence model to accurately predict the binding energy of adsorbates on the catalyst surface. This is not the first application of AI to materials discovery. The SGCNN model was developed by evolving the CGCNN model, which is specialized in predicting bulk properties of solid materials, to predict surface properties of catalytic materials. However, there is a big difference between predicting bulk properties and surface properties. When you can quickly and accurately predict the surface properties of a catalyst, you can more efficiently screen for catalysts that meet the triple bottom line of material stability, performance, and cost. In fact, when developing fuel cell anode reaction catalysts using this methodology, we were able to explore the potential of nearly 3,200 ternary candidate materials in just one day, a scale that would have taken years using the density functional theory (DFT) adsorption energy simulation calculations traditionally used to predict catalyst properties. [Figure 2] Machine learning-driven material screening workflow for each anode and cathode of fuel cell The researchers developed a novel ternary (Cu-Au-Pt) alloy catalyst through experimental validation of 10 catalysts with the potential to outperform platinum catalysts out of approximately 3,200 candidate materials. The catalyst uses only 37% of the element platinum compared to pure platinum catalysts, but the kinetic current density is more than twice as high as that of pure platinum catalysts. The catalyst also exhibits excellent durability, with little degradation after 5,000 stability tests. "In the future, we plan to continue to build high-quality adsorption energy data and perform more sophisticated AI modeling, which will further improve the success rate of catalytic material development," said Dr. Kim of KIST. The new methodology has the advantage of being immediately applicable not only to catalysts for hydrogen fuel cells, but also to various catalytic reactions such as water electrolysis-based hydrogen production, which is essential for the realization of the hydrogen economy. The team plans to further reduce the unit cost and improve the performance of the developed catalysts through material and system optimization. ### KIST was established in 1966 as the first government-funded research institute in Korea. KIST now strives to solve national and social challenges and secure growth engines through leading and innovative research. For more information, please visit KIST’s website at https://eng.kist.re.kr/ The research was supported by the Samsung Future Technology Fostering Project (SRFC-MA1801-03) of Samsung Electronics (CEO Kye-hyun Kyung) and the Materials Research Data Platform Project of the Ministry of Science and ICT (Minister Jong-ho Lee), and was published in the international journal Applied Catalysis B: Environmental. Journal : Applied Catalysis B: Environmental Title : Machine learning filters out efficient electrocatalysts in the massive ternary alloy space for fuel cells Publication Date : 24-July-2023 DOI :https://doi.org/10.1016/j.apcatb.2023.123128
- 72
- WriterDr. Kim, Donghun
- 작성일2023.10.18
- Views590
-
71
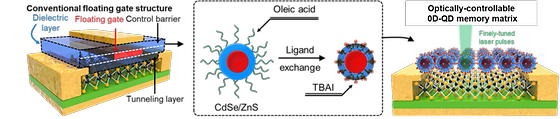
Ushering in the era of light-powered 'multi-level memories'
- KIST Developed technology to store and manipulate electronic states in quantum dots measuring 10 nanometers or less. We live in an era of data deluge. The data centers that are operated to store and process this flood of data use a lot of electricity, which has been called a major contributor to environmental pollution. To overcome this situation, polygonal computing systems with lower power consumption and higher computation speed are being researched, but they are not able to handle the huge demand for data processing because they operate with electrical signals, just like conventional binary computing systems. The Korea Institute of Science and Technology (KIST, President Seok Jin Yoon) announced that Dr. Do Kyung Hwang of the Center for Opto-Electronic Materials & Devices and Professor Jong-Soo Lee of the Department of Energy Science & Engineering at Daegu Gyeongbuk Institute of Science and Technology (DGIST, President Young Kuk) has jointly developed a new zero-dimensional and two-dimensional (2D-0D) semiconductor artificial junction material and observed the effect of a next-generation memory powered by light. Transmitting data between the computing and storage parts of a multi-level computer using light rather than electrical signals can dramatically increase processing speed. [Figure 1] 2D-0D HYBRID OPTICAL MEMORY DEVICES The research team has fabricated a new 2D-0D semiconductor artificial junction material by joining quantum dots in a core-shell structure with zinc sulfide (ZnS) on the surface of cadmium selenide (CdSe) and a molybdenum sulfide (MoS2) semiconductor. The new material enables the storage and manipulation of electronic states within quantum dots measuring 10 nm or less. When light is applied to the cadmium selenide core, a certain number of electrons flow out of the molybdenum sulfide semiconductor, trapping holes in the core and making it conductive. The electron state inside cadmium selenide is also quantized. Intermittent light pulses trap electrons in the electron band one after the other, inducing a change in the resistance of the molybdenum sulfide through the field effect, and the resistance changes in a cascading manner depending on the number of light pulses. This process makes it possible to divide and maintain more than 0 and 10 states, unlike conventional memory, which has only 0 and 1 states. The zinc sulfide shell also prevents charge leakage between neighboring quantum dots, allowing each single quantum dot to function as a memory. [Figure 2] Electron micrographs of the 2D-0D hybrid surface implemented in this study (top left), memory characteristics generated by light pulses (top right), and polynomial memory characteristics generated by multiple light pulses (bottom). While quantum dots in conventional 2D-0D semiconductor artificial junction structures simply amplify signals from light sensors, the team's quantum dot structure perfectly mimics the floating gate memory structure, confirming its potential for use as a next-generation optical memory. The researchers verified the effectiveness of the polynomial memory phenomenon with neural network modeling using the CIFAR-10 dataset and achieved a 91% recognition rate. Dr. Hwang of KIST said, "The new multi-level optical memory device will contribute to accelerating the industrialization of next-generation system technologies such as artificial intelligence systems, which have been difficult to commercialize due to technical limitations arising from the miniaturization and integration of existing silicon semiconductor devices." ### KIST was established in 1966 as the first government-funded research institute in Korea. KIST now strives to solve national and social challenges and secure growth engines through leading and innovative research. For more information, please visit KIST’s website at https://eng.kist.re.kr/ This research was supported by the Ministry of Science and ICT (Minister Jong-ho Lee) as a mid-career researcher project and a major project of KIST, and the results were published in the international journal Advanced Materials (IF: 29.4). Journal : Advanced Materials Title : Probing optical multi-level memory effects in single core-shell quantum dots and application through 2D-0D hybrid inverters Publication Date : 19-July-2023 DOI :https://doi.org/10.1002/adma.202303664
- 70
- WriterDr. Hwang, Do Kyung
- 작성일2023.10.17
- Views531
-
69
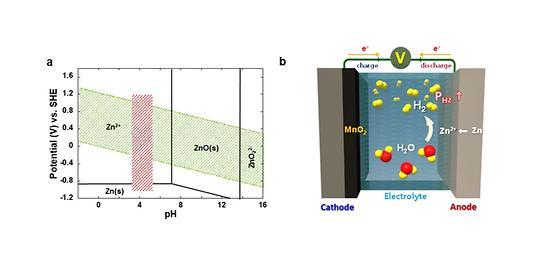
A cheaper, safer alternative to lithium-ion batteries: aqueous rechargeable batteries.
- Automatic conversion of hydrogen gas into water makes batteries safer - A breakthrough technology for the commercialization of cheaper, safer aqueous rechargeable batteries This summer, the planet is suffering from unprecedented heat waves and heavy rainfalls. Developing renewable energy and expanding associated infrastructure has become an essential survival strategy to ensure the sustainability of the planet in crisis, but it has obvious limitations due to the volatility of electricity production, which relies on uncertain variables like labile weather conditions. For this reason, the demand for energy storage systems (ESS) that can store and supply electricity as needed is ever-increasing, but lithium-ion batteries (LIBs) currently employed in ESS are not only highly expensive, but also prone to potential fire, so there is an urgent need to develop cheaper and safer alternatives. A research team led by Dr. Oh, Si Hyoung of the Energy Storage Research Center at the Korea Institute of Science and Technology (KIST) has developed a highly safe aqueous rechargeable battery that can offer a timely substitute that meets the cost and safety needs. Despite of lower energy density achievable, aqueous rechargeable batteries have a significant economic advantage as the cost of raw materials is much lower than LIBs. However, inveterate hydrogen gas generated from parasitic water decomposition causes a gradual rise in internal pressure and eventual depletion of the electrolyte, which poses a sizeable threat on the battery safety, making commercialization difficult. [Figure 1] CAUSES OF HYDROGEN GENERATION AND INCESSANT ACCUMULATION WITHIN THE CELL IN THE AQUEOUS RECHARGEABLE BATTERIES Until now, researchers have often tried to evade this issue by installing a surface protection layer that minimizes the contact area between the metal anode and the electrolyte. However, the corrosion of the metal anode and accompanying decomposition of water in the electrolyte is inevitable in most cases, and incessant accumulation of hydrogen gas can cause a potential detonation in long-term operation. [Figure 2] Proposed strategy for securing safety of the aqueous rechargeable batteries via water-regeneration To cope with this critical issue, the research team has developed a composite catalyst consisting of manganese dioxide and palladium, which is capable of automatically converting hydrogen gas generated inside the cell into water, ensuring both the performance and safety of the cell. Manganese dioxide does not react with hydrogen gas under normal circumstances, but when a small amount of palladium is added, hydrogen is readily absorbed by the catalysts, being regenerated into water. In the prototype cell loaded with the newly developed catalysts, the internal pressure of the cell was maintained well below the safety limit, and no electrolyte depletion was observed. [Figure 3] Role of composite catalysts in activating water-regeneration chemical reaction The results of this research effectively solves one of the most concerning safety issues in the aqueous batteries, making a major stride towards commercial application to ESS in the future. Replacing LIBs by cheaper and safer aqueous batteries can even trigger a rapid growth of global market for ESS. "This technology pertains to a customized safety strategy for aqueous rechargeable batteries, based on the built-in active safety mechanism, through which risk factors are automatically controlled." said Dr. Oh, Si Hyoung of KIST. "Moreover, it can be applied to various industrial facilities where hydrogen gas leakage is one of major safety concerns (for instance, hydrogen gas station, nuclear power plant etc) to protect public safety." ### KIST was established in 1966 as the first government-funded research institute in Korea. KIST now strives to solve national and social challenges and secure growth engines through leading and innovative research. For more information, please visit KIST’s website at https://eng.kist.re.kr/ This research was supported by the Ministry of Science and ICT (Minister Lee Jong-ho) through the Nano Future Material Source Technology Development Project and the Mid-Career Researcher Support Project, and the results were published on August 1 in the international journal Energy Storage Materials (IF 20.4). Journal : Energy Storage Materials Title : Highly safe aqueous rechargeable batteries via electrolyte regeneration using Pd-MnO2 catalytic cycle Publication Date : 1-August-2023 DOI :https://doi.org/10.1016/j.ensm.2023.102881
- 68
- WriterDr. Oh, Si Hyoung
- 작성일2023.10.16
- Views1124
-
67
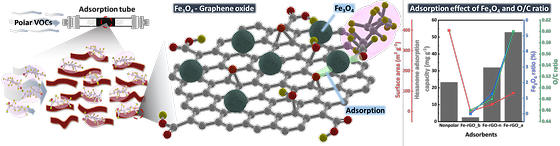
New technology for customized air purification of toxic gases
- KIST's iron oxide-graphene oxide heterostructure improves removal efficiency of harmful volatile organic compounds by up to 15 times Volatile organic compounds (VOCs) in daily products such as paints, adhesives, furniture, cosmetics, and deodorants make our lives easier. However, constant exposure can cause serious health problems such as respiratory illness, headaches, dermatitis, and cancer. Natural ventilation is the most effective way to reduce VOCs in indoor air, but recently, air purifiers have become a common method to maintain indoor air quality due to the frequent extreme outdoor condition (e.g. high concentration of fine dust, heat waves, and extreme cold). Generally, air purifiers remove VOCs by adsorption using activated carbon, which has a non-polar carbon surface and a large specific surface area. This activated carbon can effectively remove non-polar substances such as toluene and benzene, but cannot remove polar substances such as ketones and aldehydes. The Korea Institute of Science and Technology (KIST, President Seok Jin Yoon) announced that Dr. Jiwon Lee and Dr. Youngtak Oh from the Center for Sustainable Environment Research have developed a new adsorbent technology that can efficiently adsorb amphiphilic VOCs, which have both hydrophilic and hydrophobic properties and are difficult to remove with existing activated carbon technology. [Figure 1] ADSORPTION MECHANISM AND ADSORPTION PERFORMANCE GRAPH OF IRON OXIDE GRAPHENE ADSORBENT FOR POLAR VOCS The KIST research team synthesized a graphene-iron oxide heterostructure by precisely controlling the surface oxidation of graphite and iron, resulting in a high adsorption capacity for amphiphilic VOCs due to the increase of oxygen functional groups and iron oxide on the surface. This unique adsorbent showed up to 15 times better adsorption efficiency for amphiphilic VOCs than conventional activated carbon adsorbents. They also found that precise oxygen functional groups and iron oxides control of the adsorbent can offer flexible surface optimization freedom for a desirable nature of the pollutant. By testing four different ketones that are difficult to control with activated carbon adsorbents, the researchers found the correlation between the length of carbon chains and the adsorption efficiency; by optimizing the content of oxygen functional groups and iron oxides in the adsorbent, they were able to bring the maximum removal efficiency for the ketones. The researchers also analyzed the sub-nanometer electron transfer phenomenon between the adsorbent and VOC molecules; they found a link between the geometric shape of the pollutant and its adsorption trend for the first time. This is expected to enable the development of customized detection and control technologies for various air pollutants in our environment. "Unlike previous studies that focused on mere improvement of the adsorption performance and regeneration efficiency of adsorbents, we succeeded in developing a breakthrough material that exceeds the limits of existing adsorbents using accessible materials such as graphite and iron, which have high commercialization potential," said Dr. Jiwon Lee. ### KIST was established in 1966 as the first government-funded research institute in Korea. KIST now strives to solve national and social challenges and secure growth engines through leading and innovative research. For more information, please visit KIST’s website at https://eng.kist.re.kr/ The research, which was conducted as a major project of KIST (Air Environment Research Program) with support from the Ministry of Science and ICT (Minister Jong-ho Lee), was published on October 1 in the Chemical Engineering Journal. Journal : Chemical Engineering Journal Title : Effect of adsorbate geometry and hydrogen bonding on the enhanced adsorption of VOCs by an interfacial Fe3O4?rGO heterostructure Publication Date : 9-August-2023 DOI : https://doi.org/10.1016/j.cej.2023.145346
- 66
- WriterDr. Lee, Jiwon, Dr. Oh, Youngtak
- 작성일2023.10.12
- Views381
-
65
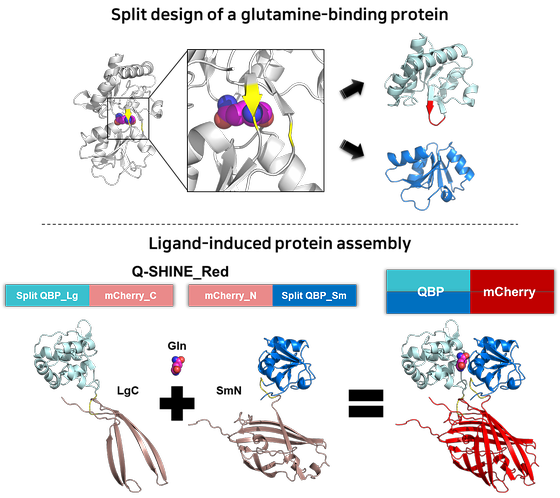
Make diagnosing serious geriatric diseases as easy as measuring blood sugar
- Developed a new biosensor for measuring glutamine with greater convenience and accuracy than before - Successfully monitored real-time changes in the concentration of glutamine in living cells In 2023, life expectancy in Korea will be 83.6 years, the third highest among OECD countries, and it is steadily increasing every year. As the proportion of the elderly population increases, the social cost of treating various geriatric diseases is also increasing rapidly, and there is a growing interest in early diagnosis of diseases. Among the various diagnostic methods, researchers are actively conducting research on measuring glutamine as an indicator of geriatric diseases by finding that the concentration of glutamine in the cells and blood of patients with serious diseases such as cancer, diabetes, and dementia is significantly changed compared to normal people. Dr. Seo, Moon-Hyeong of the Natural Product Research Center at the Korea Institute of Science and Technology (KIST), together with Dr. Park, Keunwan of the Natural Product Informatics Research Center, have developed a technology that can quickly and accurately measure glutamine concentrations without complicated measurement processes and expensive analytical equipment through the principle of 'ligand-induced protein assembly'. [Figure 1] SCHEMATIC OF Q-SHINE SENSOR DEVELOPMENT BASED ON THE PRINCIPLE OF 'LIGAND-INDUCED PROTEIN ASSEMBLY' THROUGH SPLIT AND STABILIZATION DESIGN OF A GLUTAMINE-BINDING PROTEIN Glutamine is an amino acid in the blood that is used by cells to synthesize proteins or as an energy source, and its rapid fluctuation in certain situations makes it a useful biomarker for the treatment and early diagnosis of disease. For this reason, researchers are actively studying glutamine metabolism in the body to diagnose metabolic and degenerative diseases, including cancer treatment by inhibiting the metabolism of glutamine, which is also a nutrient for cancer cells. Until now, the measurement of glutamine concentration in the body has relied on expensive specialized analytical equipment such as amino acid analyzers, which cannot measure changes in glutamine concentration in living cells in real time. In the case of relatively low-cost research kits, cumbersome pre-treatment processes such as protein removal in biological samples were required, resulting in long measurement times and low accuracy. The team developed a sensor protein for measuring glutamine based on the principle of "ligand-induced protein assembly" that can easily measure the concentration of glutamine in the blood. By separating a glutamine binding protein into two artificial proteins and then binding to the sample, and named it Q-SHINE by combining Q, the symbol for glutamine, and SHINE, which means brightly glowing. Experiments showed that the Q-SHINE sensor was highly selective, not responding to amino acids with similar structure such as glutamic acid and D-glutamine. The lowest concentration of glutamine that can be measured is 1 micromolar (µM, one millionth of a molar), which is 20 times lower than the enzymatic assay most commonly used in research kits. In addition, the sensor protein can be easily produced in E. coli, making it possible for a research kit to analyze glutamine concentrations at the same level as analytical instruments worth hundreds of millions of dollars. [Figure 2] Glutamine concentration measurement results using the Q-SHINE sensor The team also used the Q-SHINE sensor to monitor changes in glutamine concentration in the cytoplasm and mitochondria of living cells in real time. In particular, by verifying the difference in glutamine concentration between cancer cells and normal cells, it is expected to speed up the development of anticancer drugs by inhibiting glutamine metabolism. "The Q-SHINE sensor developed by KIST will enable easy monitoring of glutamine concentration, similar to the self-monitoring of blood glucose by diabetics," said Dr. Seo, Moon-Hyeong. "If used for glutamine metabolism research, it will greatly contribute to early diagnosis and identification of causes of severe geriatric diseases such as cancer, diabetes, and dementia, as well as development of cancer drugs that regulate glutamine metabolism." ### KIST was established in 1966 as the first government-funded research institute in Korea. KIST now strives to solve national and social challenges and secure growth engines through leading and innovative research. For more information, please visit KIST’s website at https://eng.kist.re.kr/ The research, which was supported by the Ministry of Science and ICT (Minister Lee Jong-ho) through the KIST Major Project and the Korea Research Foundation's Excellent New Research Project, was published in the latest issue of the international journal Sensors and Actuators, B: Chemical (IF=8.4, top 0.8% in JCR). Journal : Sensors and Actuators: B. Chemical Title : Q-SHINE: A versatile sensor for glutamine measurement via ligand-induced dimerization Publication Date : 1-September-2023 DOI : https://doi.org/10.1016/j.snb.2023.133951
- 64
- WriterDr. Seo, Moon-Hyeong
- 작성일2023.10.11
- Views302
-
63
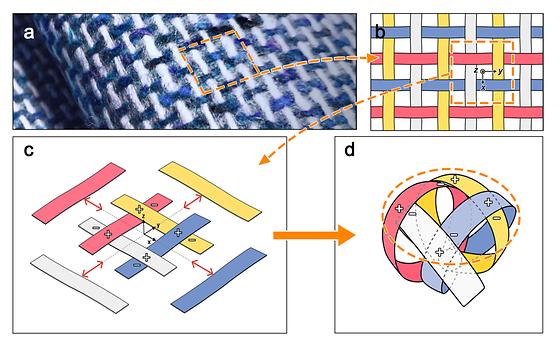
A 130g soft robot gripper lifts 100kg?
- Developing a soft robotic gripper that mimics a woven structure - Achieve high performance, cost-effectiveness, and process efficiency in a soft robotic gripper Utilizing soft, flexible materials such as cloth, paper, and silicone, soft robotic grippers is an essential device that acts like a robot's hand to perform functions such as safely grasping and releasing objects. Unlike conventional rigid material grippers, they are more flexible and safe, and are being researched for household robots that handle fragile objects such as eggs, or for logistics robots that need to carry various types of objects. However, its low load capacity makes it difficult to lift heavy objects, and its poor grasping stability makes it easy to lose the object even under mild external impact. [Figure 1] GRIPPER SCHEMATIC WITH WEAVE STRUCTURE Dr. Song, Kahye of the Intelligent Robotics Research Center at the Korea Institute of Science and Technology (KIST), along with Professor Lee, Dae-Young of the Department of Aerospace Engineering at the Korea Advanced Institute of Science and Technology (KAIST), have jointly developed a soft gripper with a woven structure that can grip objects weighing more than 100 kg with 130 grams of material. To increase the loading capacity of the soft robot gripper, the research team applied a new structure inspired by textiles, as opposed to the conventional method of developing new materials or reinforcing the structure. The weaving technique they focused on involves tightly intertwining individual threads to create a strong fabric, which can reliably support heavy objects and has been used for centuries in clothing, bags, and industrial textiles. The team used thin PET plastic The grippers were designed to allow the strips to intertwine and unwind into a woven structure. [Figure 2] Gripper behavior and performance The resulting woven gripper weighs 130 grams and can grip an object weighing 100 kilograms. Conventional grippers of the same weight can lift no more than 20 kilograms at most, and considering that a gripper that can lift the same weight weighs 100 kilograms, the team succeeded in increasing the load capacity relative to its own weight. Also, the soft robot gripper developed by the research team uses plastic, which costs only a few thousand won per unit of material, and can be used as a universal gripper that can grip objects of various shapes and weights, making it highly competitive in price. In addition, since the soft robot gripper can be manufactured by simply fastening a plastic strip, the manufacturing process can be completed in less than 10 minutes, and it is easy to replace and maintain, so the process efficiency is excellent. [Figure 3] Comparison of gripper weight to payload (maximum weight the robot can lift) for the woven gripper, soft gripper, and rigid gripper In addition to PET, which is the main material used by the research team, the gripper can also be made of various materials such as rubber and compounds that possess elasticity, allowing the team to customize and utilize grippers suitable for industrial and logistics sites that require strong gripping performance or various environments that need to withstand extreme conditions. "The woven structure gripper developed by KIST and KAIST has the strengths of a soft robot but can grasp heavy objects at the level of a rigid gripper," said Dr. Song. It can be manufactured in a variety of sizes, from coins to cars, and can grip objects of various shapes and weights, from thin cards to flowers, so it is expected to be used in fields such as industry, logistics, and housework that require soft grippers." ### KIST was established in 1966 as the first government-funded research institute in Korea. KIST now strives to solve national and social challenges and secure growth engines through leading and innovative research. For more information, please visit KIST’s website at https://eng.kist.re.kr/ KAIST is the first and top science and technology university in Korea. KAIST has been the gateway to advanced science and technology, innovation, and entrepreneurship, and our graduates have been key players behind Korea’ innovations. KAIST will continue to pursue advances in science and technology as well as the economic development of Korea and beyond. (https://www.kaist.ac.kr/en) The research was supported by the Ministry of Science and ICT (Minister Lee Jong-ho) through the KIST Major Project and the Korea Research Foundation Basic Research Program, the Overseas Advanced Scientist Invitation Program, and the Basic Research Laboratory Support Program. The results of the study were published on August 2 in the international journal Nature Communications (IF:16.6, top 8.2% in JCR) and were selected as Editors' Highlights, which introduces the best 50 papers in each field. Journal : Nature Communications Title : Grasping through dynamic weaving with entangled closed loops Publication Date : 2-August-2023 DOI : https://doi.org/10.1038/s41467-023-40358-y
- 62
- WriterDr. Song, Kahye
- 작성일2023.10.06
- Views638
-
61
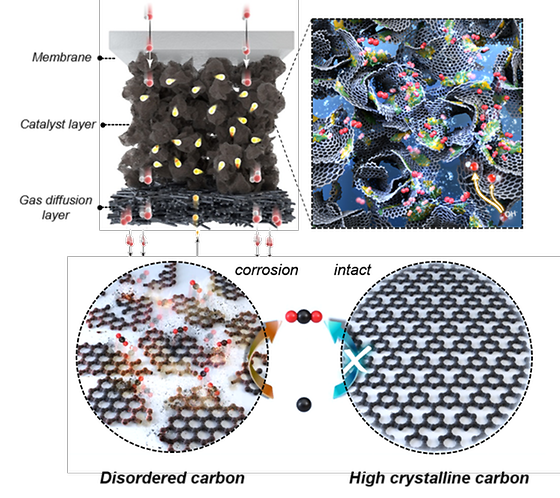
Dramatically lower the cost of producing green hydrogen.
- Suggested use of carbon in water electrolysis, which has been neglected due to corrosion issues - Using carbon supports and low-cost catalysts enables superior electrolysis performance and durability According to the International Energy Agency (IEA), global hydrogen demand is expected to reach 530 million tons in 2050, a nearly six-fold increase from 2020. Currently, the primary method of hydrogen production involves the reaction of natural gas and water vapor, resulting in what is known as 'gray hydrogen' due to its carbon dioxide emissions, constituting around 80% of total hydrogen production. In contrast, green hydrogen is produced through water electrolysis using electricity, without emitting carbon dioxide. However, a challenge lies in the inevitable use of expensive precious metal catalysts, such as iridium oxide. [Figure 1] Image of nickel-iron-cobalt layered double hydroxide supported on hydrophobic crystalline carbon and image of crystalline carbon A research team led by Dr. Yoo Sung Jong of the Hydrogen and Fuel Cell Research Center at the Korea Institute of Science and Technology (KIST) have succeeded in significantly reducing the cost of green hydrogen production by implementing an anion exchange membrane water electrolysis device with excellent performance and durability by introducing a carbon support. Carbon supports have been utilized as supports for various electrocatalysts due to their high electrical conductivity and specific surface area, but their usage has been limited because they readily oxidize to carbon dioxide in water electrolysis conditions, specifically at high voltages and in the presence of water. [Figure 2] Time-dependent-lapse transmission electron micrograph images of nickel-iron-cobalt layered double hydroxide synthesis on carbon support, high resolution scanning TEM and EDS elemental mapping images The team synthesized a nickel-iron-cobalt layered double hydroxide material, a significantly cheaper alternative to iridium, on a hydrophobic carbon support and used it as an electrocatalyst for the oxygen evolution reaction in anion exchange membrane electrolysis. The catalyst showed excellent durability due to the layered structure facing a hydrophobic carbon support and a nickel-iron-cobalt layered double hydroxide catalyst. In terms of carbon corrosion, it was found that the generation of carbon dioxide during the corrosion process was reduced by more than half, primarily because of decreased interaction with water, a key factor in carbon corrosion. It was found that the carbon dioxide generated during the corrosion process was less than half due to the reduced interaction with water, which causes corrosion of carbon. [Figure 3] Electrochemical activity evaluation of nickel-iron-cobalt layered double hydroxide and single cell test results As a result of performance evaluation, it is found that the newly developed supported catalyt achieved a current density of 10.29 A/cm-2 in the 2 V region, exceeding the 9.38 A/cm-2 current density of commercial iridium oxide. demonstrated long-term durability of about 550 hours. We also confirmed a correlation between electrolysis performance and the hydrophobicity of carbon, showing for the first time that the support's hydrophobicity can significantly affect the water electrolysis device's performance. "The results of this research confirm the applicability of water electrolysis devices on carbon supports, which have previously been limited in use due to corrosion problems, and it is expected that water electrolysis technology can grow to the next level if the research focused on catalyst development is expanded to various supports." "We will strive to develop various eco-friendly energy technologies, including green hydrogen production," said Dr. Yoo Sung Jong Yoo in KIST. ### KIST was established in 1966 as the first government-funded research institute in Korea. KIST now strives to solve national and social challenges and secure growth engines through leading and innovative research. For more information, please visit KIST’s website at https://eng.kist.re.kr/ This research was supported by the Ministry of Science and ICT (Minister Lee Jong-ho) through the KIST Major Project and Nano and Material Technology Development Project, and the Korea Energy Technology Assessment Institute(Director Kwon Ki-young) Renewable Energy Core Technology Development Project, and the results were published on August 1 in the international journal Energy & Environmental Science (IF 32.5, top 0.4% in JCR). Journal : Energy & Environmental Science Title : Realizing the Potential of Hydrophobic Crystalline Carbon as a Support for Oxygen Evolution Electrocatalysts Publication Date : 16-June-2023 DOI : https://doi.org/10.1039/d3ee00987d
- 60
- WriterDr. Yoo Sung Jong
- 작성일2023.10.04
- Views547
-
59
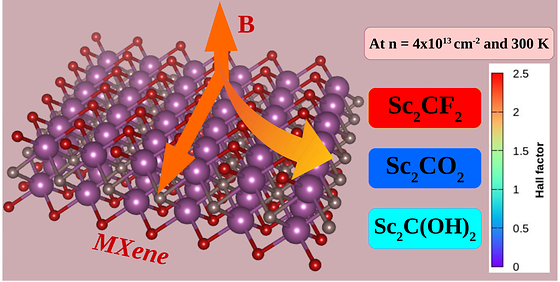
MXene, a dream new material, paves the way for mass production
- IKST researchers develop analysis model using magnetic transport characteristics of molecules attached to the surface of MXene - Establishment of property prediction and classification system is expected to be utilized to produce uniform quality MXene Developed in 2011, MXene is a two-dimensional nanomaterial with alternating metal and carbon layers, which has high electrical conductivity and can be combined with various metal compounds, making it a material that can be utilized in various industries such as semiconductors, electronic devices, and sensors. To properly utilize MXene, it is important to know the type and amount of molecules covered on the surface, and if the molecules covered on the surface are fluorine, the electrical conductivity of decreases and the efficiency of electromagnetic wave shielding decreases. However, since it is only 1 nm (nanometer - billionth of a meter) thick, it takes several days to analyze the molecules on the surface even with a high-performance electron microscope, so mass production has been impossible until now. The research team led by Seung-Cheol Lee, director of the Indo-Korea Science and Technology Center(IKST) at the Korea Institute of Science and Technology(KIST), has developed a method to predict the distribution of molecules on the surface using the magnetoresistance property of MXene. By utilizing this method, it is possible to measure the molecular distribution of MXene with a simple measurement, enabling quality control in the production process, which is expected to open the way to mass production that was not possible until now. [Figure 1] PREDICTED HALL SCATTERING FACTOR FOR MAXIN The research team developed a two-dimensional material property prediction program based on the idea that electrical conductivity or magnetic properties change depending on the molecules attached to the surface. As a result, they calculated the magnetic transport properties of MXene and succeeded in analyzing the type and amount of molecules adsorbed on the surface of MXene at atmospheric pressure and room temperature without any additional devices. By analyzing the surface of the MXene with the developed property prediction program, it was predicted that the Hall scattering factor, which affects magnetic transport, changes dramatically depending on the type of surface molecules. The Hall Scattering Factor is a physical constant that describes the charge-carrying properties of semiconductor materials, and the team found that even when the same MXene was prepared, the Hall Scattering Factor had a value of 2.49, the highest for fluorine, 0.5 for oxygen, and 1 for hydroxide, allowing them to analyze the distribution of the molecules. The Hall scattering coefficient has different applications based on the value of 1. If the value is lower than 1, it can be applied to high-performance transistors, high-frequency generators, high-efficiency sensors, and photodetectors, and if the value is higher than 1, it can be applied to thermoelectric materials and magnetic sensors. Considering that the size of the maxin is a few nanometers or less, the size of the applicable device and the amount of power required can be dramatically reduced. "Unlike previous studies that focused on the production and properties of pure MXene, this study is significant in that it provides a new method for surface molecular analysis to easily classify manufactured MXene," said Seung-Cheol Lee, director of IKIST. "By combining this result with experimental studies, we expect to be able to control the production process of MXene, which will be used to mass produce MXene with uniform quality." IKST was established in 2010 and conducts research in the areas of theory, source code, and software for computational science. In particular, source code is a programming language that implements algorithms that can be modeled and simulated, and is considered an original research in the field of computational science, and the center conducts collaborative research with Indian universities and research institutes such as IIT Bombay to develop source code. ### KIST was established in 1966 as the first government-funded research institute in Korea. KIST now strives to solve national and social challenges and secure growth engines through leading and innovative research. For more information, please visit KIST’s website at https://eng.kist.re.kr/ This research, which was conducted as a KIST Major Project (2Z06950) funded by the Ministry of Science and ICT (Minister Lee Jong-ho), was selected as a Notable Article of the Year (2023 Hot Article Collection) of Nanoscale, an international journal in the field of nanoscience (IF:6.7, JCR top 16.7%) for its originality and scalability, and was published on June 28. Journal : Nanoscale Title : Can magneto-transport properties provide insight into the functional groups in semiconducting MXenes? Publication Date : 28-June-2023 DOI :https://doi.org/10.1039/d2nr06409j
- 58
- WriterDr. Lee Seung-Cheol
- 작성일2023.09.18
- Views503
-
57
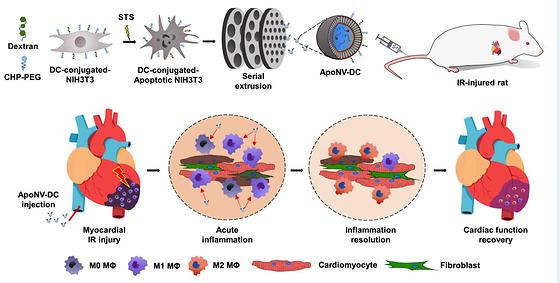
Myocardial infarction, the number one cause of sudden death, may be treated by modulating the immune response.
- Reduced inflammation at the site of myocardial infarction and improved heart function demonstrated - Novel therapy to modulate immune response with apoptotic cell-derived nanovesicles Myocardial infarction, the number one cause of sudden death in adults and the number two cause of death in Korea, is a deadly disease with an initial mortality rate of 30%, and about 5-10% of patients die even if they are transported to a medical center for treatment. The number of myocardial infarction patients in Korea has been increasing steeply, from 99,647 in 2017 to 126,342 in 2021, an increase of 26.8% in five years. Until now, drug administration, percutaneous angioplasty, and arterial bypass surgery have been known as treatments, but they are difficult to apply to severe cases that do not respond to them. Dr. Yoon Ki Joung and Dr. Juro Lee of the Biomaterials Research Center at the Korea Institute of Science and Technology (KIST), together with Prof. Hun-Jun Park and Dr. Bong-Woo Park of the Catholic University of Korea College of Medicine, have developed a new treatment for myocardial infarction that uses nanovesicles derived from fibroblasts with induced apoptosis to modulate the immune response. [Figure 1] SCHEMATIC ILLUSTRATION OF TREATMENT OF MYOCARDIAL ISCHEMIA-REPERFUSION (IR) INJURY WITH THE TARGETED DELIVERY OF APONV-DCS Myocardial infarction is an ischemic heart disease in which the coronary arteries, the blood vessels that supply blood to the heart, become narrowed or blocked, resulting in insufficient blood supply to the heart muscle, which causes nutrient and oxygen deficiency in the myocardium, leading to poor heart function. According to market research firm Technavio, the global myocardial infarction therapeutics market is expected to reach $2.02 billion by 2026, at a CAGR of 4.7%. In recent years, stem cell-derived nanovesicles, such as exosomes, have been used to treat myocardial infarction by modulating the inflammatory response, but stem cells are difficult to produce in large quantities, limiting their economic viability. [Figure 2] Effect on viable myocardium and fibrosis area at 4 weeks after treatment. The research team identified the possibility of treating severe myocardial infarction by reducing the inflammatory response in the heart muscle through a nanomedicine based on apoptotic cells, which are cells that commit suicide due to biochemical changes in their cells. This response was achieved by attaching peptides specific to the site of ischemic myocardial infarction and substances specific to macrophage phagocytosis to the surface of fibroblasts. To this end, the team developed anti-inflammatory nanovesicles that can be delivered specifically to macrophages at the site of myocardial infarction. [Figure 3] Prevention of cardiac function deterioration 4 weeks after the ApoNV-DCs injection. In animal studies, we found that intravenously injected nanovesicles were effectively delivered to the myocardial infarction site in rats and were specifically recruited to macrophages. As a result, the left ventricular ejection fraction, an indicator of the contractile force of the left ventricle, increased by more than 1.5 times compared to the control group for 4 weeks. In addition, the effects of reducing inflammation and fibrosis, and increasing blood vessels preservation rate enhanced cardiomyocytes survival, which resulted in cardiac function improvement. "This is the first study to use nanovesicles produced from apoptosis-induced cells to treat myocardial infarction, and it has the advantage of being able to mass-produce them because it uses other cells rather than stem cells," said Dr. Yoon Ki Joung of KIST. "In the future, we plan to conduct a research to verify the effectiveness and safety of the treatment, including clinical trials, through a collaborative research with Catholic University of Korea Medical School and bio companies." ### KIST was established in 1966 as the first government-funded research institute in Korea. KIST now strives to solve national and social challenges and secure growth engines through leading and innovative research. For more information, please visit KIST’s website at https://eng.kist.re.kr/ This research was supported by the Ministry of Science and ICT (Minister Lee Jong-ho) through the Korea Research Foundation Nano and Material Technology Development Project and the Sejong Science Fellowship Program, and the results were published in the June issue of Advanced Functional Materials (IF:19.0, JCR top 4.7%), an international journal in the field of materials. Journal : Advanced Functional Materials Title : Targeted Delivery of Apoptotic Cell-Derived Nanovesicles prevents Cardiac Remodeling and Attenuates Cardiac Function Exacerbation Publication Date : 02-June-2023 DOI : https://doi.org/10.1002/adfm.202210864
- 56
- WriterDr.Joung, Yoon Ki
- 작성일2023.08.25
- Views395
-
55
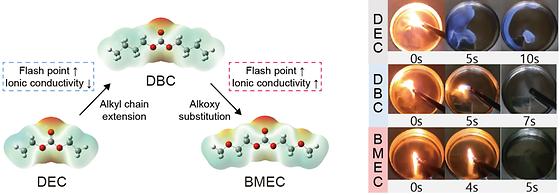
Developing a nonflammable electrolyte to prevent thermal runaway in lithium-ion batteries
- Tailoring the molecular structure of organic carbonates in commercial electrolytes reduces the fire hazard of batteries - Nonfluorinated, nonflammable electrolytes present a viable route to achieving thermally stable high-performance batteries The Korea Institute of Science and Technology(KIST, President Seok-Jin Yoon) announced that a collaborative research team led by Dr. Minah Lee of the Energy Storage Research Center, Professor Dong-Hwa Seo of the Korea Institute of Science and Technology(KAIST), and Drs. Yong-Jin Kim and Jayeon Baek of the Korea Institute of Industrial Technology(KITECH) has developed a nonflammable electrolyte that does not catch fire at room temperature by tailoring the molecular structure of linear organic carbonate to prevent fire and thermal runaway in lithium-ion batteries. As the use of medium and large-scale lithium-ion batteries in electric vehicles and energy storage systems(ESS) expands, concerns about fires and explosions are growing. Fires in batteries occur when batteries are short-circuited due to external impacts, abuse or aging, and the thermal runaway phenomenon accompanied by a serial exothermic reactions makes it difficult to extinguish the fire and poses a high risk of personal injury. In particular, the linear organic carbonate used in commercial electrolytes for lithium-ion batteries has a low flash point and easily catches fire even at room temperature, which is a direct cause of ignition. [Figure 1] MOLECULAR DESIGN STRATEGY FOR HIGH-FLASHPOINT ELECTROLYTE AND COMPARISON OF ROOM TEMPERATURE IGNITION PROPERTY Until now, in order to reduce the flammability of the electrolyte, Intensive fluorination in the solvent molecules or highly concentrated salts has been widely adopted. As a result, the lithium-ion transport in the electrolyte was reduced or those were incompatible with commercial electrodes, limiting their commercialization. By simultaneously applying alkyl chain extension and alkoxy substitution to the diethyl carbonate(DEC) molecule, a typical linear organic carbonate used in commercial lithium-ion battery electrolytes, the researchers developed a new electrolyte, bis(2-methoxyethyl) carbonate(BMEC), with enhanced flash point and ionic conductivity by increasing intermolecular interactions and the solvation ability. The BMEC solution has a flash point of 121°C, which is 90°C higher than that of the conventional DEC solution, and thus is not ignitable in the temperature range for conventional battery operation. BMEC can dissociate lithium salt stronger than its simple alkylated counterpart, dibutyl carbonate(DBC), solving the problem of slower lithium ion transport when reducing flammability by increasing intermolecular interaction. As a result, it retains more than 92% of the original rate capability of the conventional electrolyte while significantly reducing the fire hazards. [Figure 2] Nail-penetration test results of 4Ah pouch cells using conventional and new electrolyte In addition, the new electrolyte alleviated 37% of combustible gas evolution and 62% of heat generation than those of the conventional electrolyte. The research team demonstrated the stable operation of 1Ah lithium-ion batteries over 500 cycles by combining the new electrolyte with a high nickel cathode and a graphite anode. They also conducted a nail-penetration test on a 70% charged 4Ah-level Li-ion battery and confirmed the suppressed thermal runaway. [Figure 3] (Left) Electrolyte of a commercial lithium-ion battery (DEC) and a new electrolyte (BMEC) developed by a joint research team from KIST, KITECH, and KAIST (right). Dr. Minah Lee of the KIST stated, "The results of this research provide a new direction for designing nonflammable electrolytes, which has been inevitably sacrificed the electrochemical property or economic feasibility." "The developed nonflammable electrolyte has cost competitiveness and excellent compatibility with high-energy density electrode materials, so it is expected to be applied to the conventional battery manufacturing infrastructure. Ultimately, it will accelerate the emergence of high-performance batteries with excellent thermal stability." Dr. Jayeon Baek of KITECH stated, "The BMEC solution developed in this research can be synthesized by transesterification using low-cost catalysts and easily scaled up. In the future, we will develop the synthesis method using C1 gas (CO or CO2) to enhance its eco-friendliness further." ### KIST was established in 1966 as the first government-funded research institute in Korea. KIST now strives to solve national and social challenges and secure growth engines through leading and innovative research. For more information, please visit KIST’s website at https://eng.kist.re.kr/ This research was supported by the the National Research Council of Science & Technology and the Mid-Career Research Progam of the National Research Foundation of Korea grant by the Korea government Ministry of Science and ICT(Minister Jong-Ho Lee). The research result was published in the latest issue of Energy & Environmental Science (IF 32.5, JCR top 0.4%), an international journal in the field of energy and environmental science. Journal : Energy & Environmental Science Title : Molecularly engineered linear organic carbonates as practically viable nonflammable electrolytes for safe Li-ion batteries Publication Date : 12-July-2023 DOI : https://doi.org/10.1039/d3ee00157a
- 54
- WriterDr. Lee, Minah
- 작성일2023.08.01
- Views573