Latest Research News
Moving Beyond the Small Hydrogen Car Era to Hydrogen Trucks and Airplanes
- Date : 22-06-29
- Views : 574
- Discovery of dispersing solvent parameters affecting ionomer microporous structure
- Performance improvement of proton-exchange membrane hydrogen fuel cells under high-temperature and non-humidified conditions
A hydrogen fuel cell, which is a device that generates electrical energy through the reaction of hydrogen and oxygen in air, is gaining increasing attention as an eco-friendly power-generating device that do not emit pollutants. Among the various hydrogen fuel cells, proton-exchange membrane fuel cells (PEMFCs), which use ion-exchangeable polymer membranes as electrolytes, are relatively lightweight and have a faster start-up time. Owing to these characteristics, they are actively studied as a power source for homes and automobiles.
Owing to their high electrochemical reaction rates and strong resistance to impurities at high operating temperatures, PEMFCs are ideal for applications in high-performance transportation, such as trucks, subways, trains, airplanes, and ships. However, a separate cooling system is required at high temperatures (>100 °C) to prevent ionic conductivity reduction triggered by evaporation in polymers. The weight added by cooling systems decreases the efficiency of PEMFCs. To use PEMFCs without a cooling system, high-temperature performance improvement and (80–200 ℃) non-humidification conditions are essential.
The Korea Institute of Science and Technology (KIST, President Seok-Jin Yoon) announced that Dr. Sung-Soo Lee’s team at the Material Structure Control Research Center, KIST, South Korea, and Dr. Yu-Seung Kim’s team at the Los Alamos Research Center (LANL), the U.S, have jointly developed a platform for controlling the microporous structure of ionomers, which is the key to improving the PEMFC performance.
When polymer-containing phosphonic acid (RPO3H2) and polymer-containing sulfonic acid (RSO3H) combine, hydrogen from the sulfonic acid, which has a higher acid strength, is transferred to the phosphonic acid, thereby forming a protonated phosphonic acid ionomer. Using such a composite ionomer enables waterless ionic conduction, resulting in an increased performance of hydrogen fuel cells, even under high-temperature and non-humidified conditions. Further performance improvement can be expected through increased usage of reactive gases, such as hydrogen and oxygen.
The KIST and LANL joint research team induced the accessibility of reactive gases by manipulating a composite ionomer to obtain a microporous structure. The team discovered the dependency of the composite ionomer’s microporous structure on the solvent in which it is dispersed, as well as a direct correlation between the dispersion solvent’s pKa (acid strength) and the phosphonic acid ionomer’s microporous structure. Subsequently, a performance evaluation of a high-temperature-hydrogen fuel cell confirmed that the composite ionomer’s microporous structure positively affected the performance of fuel cells.
Dr. Sung-Soo Lee of KIST said, “The achievement is in discovering how important ionomer dispersion solvent pKa is in high-temperature-hydrogen fuel cells.” He revealed the significance of the study, adding, “We have expanded the use of hydrogen fuel cells from small transportation to bigger mobility such as trucks and ships.”
This research was performed as part of the Advanced Research Projects Agency-Energy of the US Department of Energy, the Material Innovation Lead Project of the Ministry of Science and ICT (Minister Jong-Ho Lee), and the major projects of KIST. The research results are published in ‘ACS Energy Letters’ (IF:23.101, JCR top 3.302%).
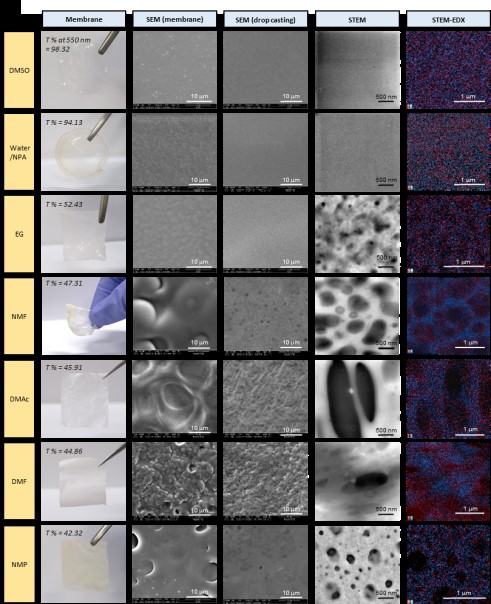
[Figure 1]
The
appearance of the protonated phosphonic acid film affected by the ionomer
dispersion solvent, and the films’ microstructures and elemental analyses
examined using electron microscopy.
The red regions denote sulfonic acid (S) groups, and blue regions denote phosphonic acid (P) groups. The visible light transmittance (T%) and STEM-EDX [A2] at 550 nm, which are related to ionomer compatibility, are shown. A higher visible light transmittance results in increased film transparency.
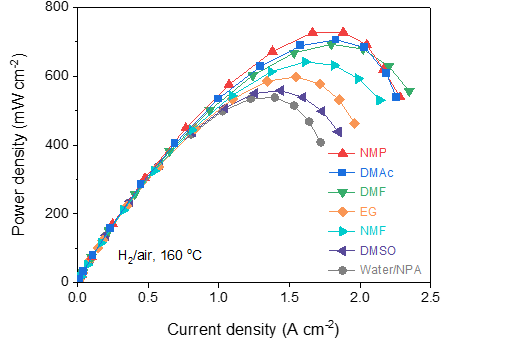
[Figure 2]
MEAs’
power density treated with various dispersion solvents.
MEA molded with a highly porous organic solvent displays a maximum power density.