Latest Research News
Development of COVID-19 AOP-Based Regulatory Toxicology Assessments
- Date : 23-06-07
- Views : 393
- First Approval of COVID-19 AOPs in Work Plan for an OECD Project
- Globally Recognized for Excellence of AOPs Published in EURL ECVAM Status Report
The EURL ECVAM Status Report describes research, dissemination and promotion activities undertaken recently by the European Commission Joint Research Centre’s EU Reference Laboratory for Alternatives to Animal Testing (EURL ECVAM) to further the uptake and use of non-animal methods and approaches in science and regulation. The principle of the Three Rs of animal use, i.e. Replacement, Reduction and Refinement, in basic, applied and translational research, as well as for regulatory testing purposes, is firmly anchored in EU legislation, with complete phasing out of animal procedures being the ultimate goal. This report provides an overview of the activities and achievements of EURL ECVAM, which is hosted by the Joint Research Centre, and covers topics such as the development, validation and dissemination of non-animal methods in testing and research. As a hands-on initiative into that direction, EURL ECVAM has been facilitating an interdisciplinary crowdsourcing project, called CIAO (Modelling the Pathogenesis of COVID-19 using AOPs). CIAO is operated on the assumption that AOPs can provide an integrative means for organizing the abundant, rapidly-evolving and dispersed knowledge on COVID-19’s pathogenesis.

[Figure 1] More than 80 scientists from 70 institutions worldwide were collaborating in the CIAO project (https://www.ciao-covid.net).
The Korea Institute of Science and Technology Europe (KIST Europe) first registered AOP 319 and 320, and then received approval on an OECD Work Plan to seek a new breakthrough in the pandemics field with the development of a reliable assessment tool for COVID-19. Since then, Dr. Youngjun Kim's research team from the Environmental Safety Group at KIST Europe has been involved with a CIAO project which can effectively evaluate the toxic effects of COVID-19 using a mechanistic understanding of the disease. More than 70 scientists from around the world were (and still are) participating in the CIAO project and developing AOPs which are able to model the COVID-19 disease process. While AOPs are widely acknowledged in chemical safety assessment and regulatory toxicology, they also have the potential to be of great value for biomedical research. The COVID-19 pandemic presented a unique opportunity to introduce the biomedical community to the AOP framework, which is not chemical-specific in nature.
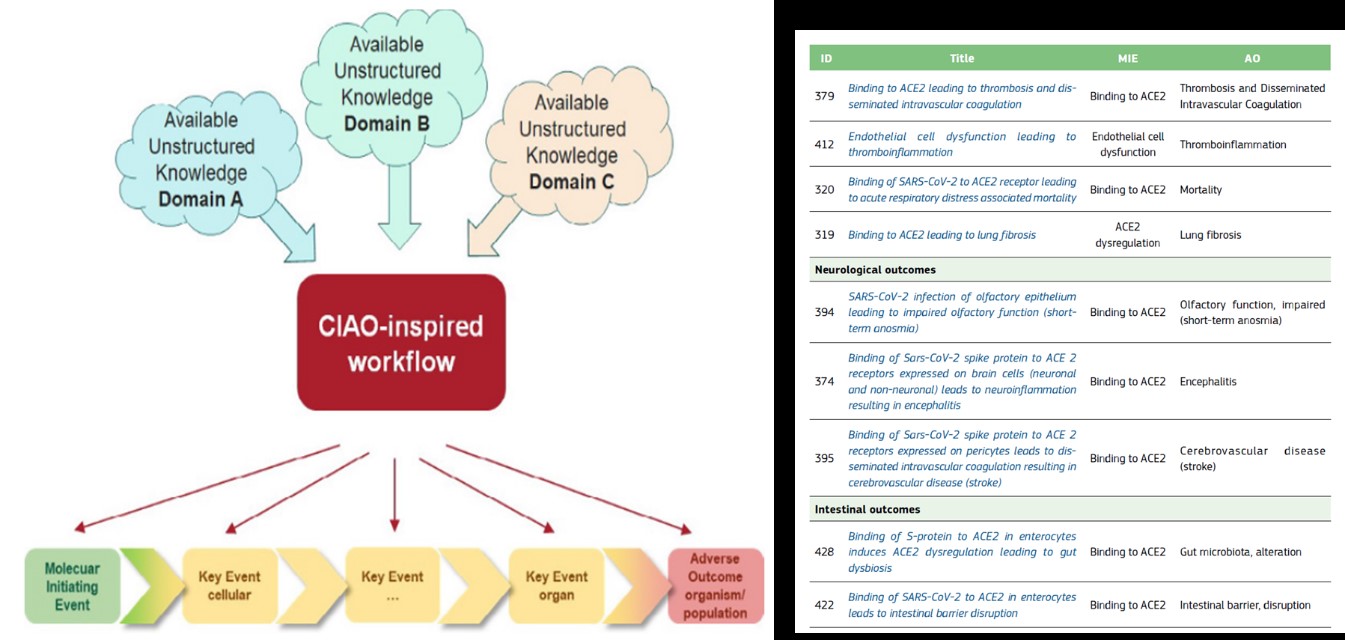
[Figure 2] COVID-19 related AOPs developed within CIAO published in EURL ECVAM policy report (OECD project 1.96).
A mechanistic understanding of the disease permits the combination of data from in vitro models for virus characterization with data from animal and human studies depicting the inflammatory response and various clinical outcomes. This AOP-based description of how the SARS-CoV-2 virus infects the body also helps to capture how various factors modulate the clinical outcomes, increasing our understanding of why some populations are more vulnerable than others. The modular aspect of AOPs also allows the development of a COVID-19-related AOP network where key biological events, interrelations between outcomes, and knowledge gaps become more evident.
In a statement, Dr. Kim mentioned, "The COVID-19 AOPs published in the EURL ECVAM Status Report represent the first trial of this kind in the world. They carry significant meaning for conventional chemical-specific AOPs since they can be expanded to include biomedical applications and linked to specific diseases, while also holding regulatory relevance in terms of non-animal testing methods."
The Joint Research Centre (JRC) has been closely involved with promoting the development of COVID-19 AOPs. It has contributed to the advancement of these non-animal testing methods through research, validation and regulation efforts such as OECD test guidelines approval and databases related to non-animal methods, which provide valuable resources for scientists, regulators and other stakeholders.
In conclusion, this report presents an overview of the newest AOPs in the area of non-animal methods. It covers topics such as in vitro methods, computational models and integrated testing strategies, providing insights into their potential applications and limitations, ultimately leading to improved scientific practices and more ethical approaches in research and regulatory toxicology.
This research was conducted as part of KIST Europe’s Significant Research Program. The research results were published online in the latest issue of the EURL ECVAM Status Report, a world-renowned policy report prepared by the Joint Research Centre (JRC), the European Commission’s science and knowledge department. (Publications Office of the European Union, 2023, ISSN 1831-9424).